Analysis of diabetes attitudes, wishes and needs in Switzerland, the Swiss DAWN2™
Study
DOI: https://doi.org/10.57187/smw.2023.40023
Oliver
Andreas Kuratli, Carolina
Gross, Markus
Laimer, Andreas
Melmer
Department of Diabetes,
Endocrinology, Nutritional Medicine and Metabolism, Inselspital, Bern University Hospital
and University of Bern, Bern, Switzerland
Summary
AIMS OF THE STUDY: Swiss DAWN2™ aimed to evaluate the difficulties and unmet
needs of individuals with diabetes and stakeholders, based on the assessments
of diabetes care and self-management: the individual burden
of disease, the perception of the quality of medical care, and the treatment
satisfaction of individuals with diabetes living in the Canton of Bern. The results
of the Swiss
cohort were analysed and compared with the global DAWN2™ results.
METHODS: 239 adult individuals with diabetes
were enrolled in a cross-sectional study at the Department of Diabetes, Endocrinology,
Nutritional Medicine and Metabolism at the
University Hospital of Bern between 2015 and 2017. The participants completed
validated online questionnaires regarding health-related quality of life
(EQ-5D-3L) and emotional distress (PAID-5), diabetes self-care activities
(SDSCA-6), treatment satisfaction (PACIC-DSF), and health-related wellbeing
(WHO-5). Eligibility criteria were as follows: participants were aged >18
years, had a diagnosis of diabetes type 1 or 2 since at least 12 months and gave written
informed
for the participation in the present study.
RESULTS: When compared globally, the Swiss
cohort reported a higher quality of life (77.28 ± 16.73 vs. 69.3 ± 17.9
EQ-5D-3L score, p <0.001) and lower emotional distress (22.28 ± 20.94 vs.
35.2 ± 24.2 PAID-5 score, p = 0.027). Higher frequencies of self-measurement of blood
glucose (6.43 ± 1.68 vs. 3.4 ± 2.8 SDSCA-6
score, p <0.001) and physical activity (4.40 ± 2.04 vs. 3.8 ± 2.7 SDSCA-6
score, p = 0.05) were reported. PACIC-DSF revealed higher satisfaction concerning
organisational aspects
of patient care (60.3 ± 1.51 vs. 47.3 ± 24.3, p<0.001) and higher
health-related well-being as compared to the global score (71.38 ± 23.31 vs. 58 ±
13.8 WHO-5 Well-Being Index, p <0.001).
HbA1c >7% correlated to
emotional distress (PAID-5, 26.08 ± 23.37 vs. 18.80 ± 17.49, p = 0.024), unfavourable
eating habits (4.28 ± 2.22 vs. 4.99 ± 2.15, p = 0.034) and decreased physical
activity (3.95 ± 2.16 vs. 4.72 ± 1.92, p = 0.014). Sleeping problems were most commonly
reported (35.6%).
In total, 28.8% of respondents completed diabetes-related
educational programs.
CONCLUSION: In global comparison, Swiss DAWN2™ showed a
lower burden of disease and yet a higher level of treatment satisfaction in
patients who were treated in
Switzerland. Further studies are required to assess the quality of diabetes
treatment and unmet needs in patients treated outside of a tertiary care center.
Introduction
The successful management of diabetes demands discipline, knowledge, and proper self-management
from patients, family members, and caregivers. These requirements pose major challenges
for patients and associates alike, leading to psychosocial distress and disease burden.
According to a recent report, exposure to distress indirectly correlates to diabetes
treatment success [1]. When left untreated, distress may contribute to decreased glycaemic
control and increase the risk for glycaemic complications, which in turn may exacerbate
psychological and mental health problems [2, 3]. Indeed, there is a higher-than-normal
prevalence of mental health conditions in individuals with diabetes [4–7]. This is
particularly true for depression, with a prevalence that is 2–3 times higher compared
to the general population [8].
Treatment-associated distress is determined by the quality and structure of medical
care, which, among other important aspects, comprise the successful acquisition and
consideration of patients’ wishes and needs [9]. To examine the interplay between
psychosocial stress and treatment quality in individuals with diabetes, the Diabetes
Attitudes, Wishes, and Needs Study (DAWN) was conducted in 2001. In a cross-national
setup including 17 countries worldwide, DAWN was aimed at evaluating the psychosocial
needs, views, and wishes of patients and stakeholders. It highlighted the link between
psychosocial stress and the prevalence of diabetes-related complications [10, 11].
The authors identified psychosocial distress and inadequate support as determinants
of poor diabetes management and highlighted the need to optimize psychosocial support
in the treatment environment of diabetes [12]. In 2013, the international DAWN2™ study
was conducted to illustrate the unmet needs of individuals with diabetes and also
healthcare providers, in order to facilitate and strengthen joint treatment efforts
[13]. DAWN2™ was performed by a multinational, multidisciplinary alliance of 17 countries
worldwide. Its results enabled a better understanding of the priorities and psychosocial
needs of individuals with diabetes. Thus, the present Swiss DAWN2™ study was designed
to extend the existing knowledge by illustrating the burden of disease, the perception
of medical care, and the treatment satisfaction of individuals with diabetes living
in Switzerland. The study also provides a comparison of its results with the former
studies conducted by the DAWN2™ consortium.
Methods
Between
March 2015 and February 2017, patients were recruited from the outpatient
clinics of the Department of Diabetes, Endocrinology, Nutritional Medicine, and
Metabolism, a tertiary care center at the University Hospital of Bern. The inclusion
criteria were:
written informed consent, age >18
years and diagnosis of type 1 or type 2 diabetes ≥12 months before
inclusion. The exclusion criteria were: diagnosis of gestational diabetes,
concomitant terminal illness, and cognitive impairments interfering with the conduction
of the study.
All
participants received access to an online platform (Research Electronic Data
Capture [RedCap]) hosted by an independent clinical trials unit at the
University of Bern. They were asked to complete questionnaires having a
cross-sectional design. These questionnaires comprised the European Quality of
Life 5 Dimensions 3 level version (EQ-5D-3L), the Problem Areas in Diabetes
Scale 5 (PAID-5), the Summary of Diabetes Self-Care Activities measure
(SDSCA-6), the Patient Assessment of Chronic Illness Care-DAWN Short Form
(PACIC-DSF), and the WHO-5 Well-Being Index (WHO-5) [14]. All surveys were reviewed
and
approved in the original English version and were translated into the primary
languages of Switzerland (German, French, and Italian).
The EQ-5D-3L
was used as a standardised measure of health status developed by the EuroQol
Group, providing a simple, generic health measure for clinical and economic
appraisal [15]. EQ-5D-3L enables a
continuous assessment of the health-related quality of life and the
self-reported health state using a scale ranging from 0 to 100.
The PAID-5 five-item
short form of the Problem Areas in Diabetes Scale (question items 3, 6, 12, 16,
19) was used to identify diabetes-related distress using a scale with a range
of 0–4. A total score ≥8 was deemed as warranting further assessments.
The SDSCA-6
questionnaire is a brief self-report measure. Using this questionnaire,
diabetes self-care-related activities were assessed briefly, with the evaluation of
specific
self-care activities performed in the past seven days (0 = no implementation on
any day of the week; 7 = implementation on every day of the week). An average
score was obtained for each dimension in a range of 0 to 7, with a higher score
suggesting better self-care. To be precise, activities such as eating habits, physical
activities, self-measurement of blood glucose, compliance with recommended treatment
strategies, and evaluation of diabetes-related health issues were assessed.
The PACIC-DSF
was used to evaluate and quantify the patients’ satisfactory states with regard to
patient-centred care in the preceding six months. In general, this
questionnaire includes 12 items evaluating the following: the effects of
diabetes on life (item 1), diabetes medications and their effects (item 2), the
patient’s opinion on the diabetes treatment plan (item 3), the encouragement of
patients to ask questions (item 4), the aspect of listening to patients (item
5), the setting of specific goals to improve diabetes treatment (item 6), the
setting of plans to achieve treatment goals (item 7), confidence conveyed by the diabetes
expert in dealing with diabetes symptoms (item 8), the aid to patients so that they
gather support from friends (item 9), the aspect of patients getting support from
family or the community (item 10), the encouragement of patients to join groups
to get help (item 11), and the task of contacting patients for information
about treatment progress and their satisfaction with treatment organisation
(item 12).
The effects of diabetes on people’s personal lives and the patients’
satisfaction with diabetes treatment organisation were highlighted and globally compared,
in detail, in this study (items 1 and 12). The score on the form used in this
study has a range of 1 to 5, with a higher score being indicative of a more patient-centred
care.
The WHO-5
Well-Being index was used to indicate the presence of depressive episodes. A
score ≤28 indicated an episode of psychological unrest and was
likely associated with the development of depression.
Self-reported
haemoglobin A1c (HbA1c) levels were separated in two groups: The first group
“improvement required” (HbA1c >7%) and second group “no improvement required”
(H bA1c <7%) were then used to evaluate the effects of HbA1c on parameters derived
from questionnaires.
Participants
and data collection
Recruitment
was initiated by treating physicians in the outpatient clinics of the Department of
Diabetes, Endocrinology,
Nutritional Medicine, and Metabolism at the University Hospital of Bern. Type
1 diabetes was defined in terms of patient age ≤30 years, with the
initial and ongoing requirement of insulin substitution at the time of
diagnosis. Type 2 diabetes was defined by patient age >30, without
initial insulin therapy. Patient demographics comprised age, sex,
date, diabetes type, treatment type, treatment duration, education, and annual
financial income. After providing written informed consent, the participants
gained access to an online platform in order to complete
the questionnaires. Missing, conflicting, and ambiguous chart elements were
coded as missing data.
After the
completion of all questions, the participants were able to announce the
completeness of the data input. If missing data fields were observed, the
respective participants were contacted by the study team and were asked to complete
the missing items, if possible. Thereafter, data were pseudonymised
and locked in the database. At the end of the recruitment period, automated
data reports were created and used for analysis.
Statistical
analysis
Mean and
standard deviation were chosen to describe the central tendency and dispersion
of continuous variables, while relative frequencies were indicated for
categorical variables. Cronbach’s Alpha was calculated to gauge scale
reliability. Chi² and t-tests were conducted to determine the statistical
significance of differences in the categorical and metric variables between
diabetes types within the study. For the comparison of this Swiss study with
the global study’s mean values, one-sample t-tests were conducted for
continuous measures. Exact binomial tests were also used for the comparison of
proportions. An α-level of 0.05 was chosen as the level of significance, and a
two-sided test was used in all analyses. To test the distribution of systemic
item variations across diabetes types, a comparison of mean values was done
using Welch’s t-test. The analysis was conducted using R version 4.2.0 [16].
Ethical
considerations
All
procedures performed in the present study involving human participants were in
accordance with the ethical standards of the institutional and/or national
research committee and with the 1964 Helsinki Declaration and its later
amendments. All participants declared their consent in writing prior to any
study-related activities. Moreover, the present study was approved by the
Regional Ethics Committee of Bern under the project number 2014-03434 and was registered
at https://clinicaltrials.gov/ with the unique trial identifier number NCT02211742. The
full trial protocol and an anonymised data report can be assessed on demand from
the corresponding author.
Results
A total of 409 eligible patients were identified, of which 239 patients completed
the online questionnaires. Of those included in the analysis, 115 participants had
type 1 diabetes and 43 had type 2 diabetes. In 81 participants, the type of diabetes
could not be assigned and was not reported. Table 1 reports the Swiss study sample,
including
sociodemographic data, diabetes duration, educational level and income range.
Table 1Baseline characteristics of the Swiss study
cohort.
| Parameter |
Overall study cohort |
Type 1 diabetes |
Type 2 diabetes |
| Number of
participants (n) |
239 |
115 |
43 |
| Age (years, ± mean) |
50.25
(16.08) |
41.60
(13.69) |
64.70
(9.74) |
| Men |
120 |
43 (37.4) |
32 (74.4) |
| Women |
117 |
71 (61.7) |
11 (25.6) |
| BMI (kg/m2) |
26.04
(4.83) |
25.46
(4.21) |
27.20
(5.52) |
| Diabetes
duration in years |
22.51 |
27.73 |
16.05 |
| Education |
No
secondary education |
57 |
26 |
12 |
| At least
secondary education |
145 |
77 |
27 |
| Annual
income in CHF |
20.000–40.000 |
16 |
10 |
3 |
| 40.000–80.000 |
70 |
35 |
12 |
| 80.000–120.000 |
63 |
28 |
14 |
| 120.000–160.000 |
17 |
10 |
4 |
| 160.000–200.000 |
4 |
0 |
2 |
| >200.000 CHF |
13 |
3 |
4 |
| N.A. |
56 |
29 |
4 |
Table 2 reports the number of participants (n) who have
completed the above-mentioned questionnaires and percentages of missing,
conflicting or incomplete/ ambiguous data.
Table 2Number of participants completing each
questionnaire. Overall study cohort: n = 239.
| Number of participants according to specific questionnaire |
n |
Missing, conflicting or incomplete/ambiguous data |
| EQ-5D-3L |
200 |
16% |
| PAID-5 |
197 |
17% |
| PACIC-DSF |
171 |
21% |
| PACIC-DSF Item 1 |
186 |
22% |
| PACIC-DSF Item 12 |
183 |
23% |
| WHO-5 |
149 |
38% |
| SDSCA-6 |
191 |
20% |
Figures 1–3
illustrate the scores of completed questionnaires in the Swiss cohort and the
comparison with the global average.

Figure 1Scores derived from
EQ-5D-3L, PAID-5, PACIC-DSF, WHO-5 in Swiss DAWN2, compared to global
average scores. Weights are based on each country, as
publicly provided by each country's survey advisory group. Sample means are
depicted as points. The red point represents the mean value of the Swiss study
results, compared to the blue dot representing the entire sample of global
study data of people with diabetes.

Figure 2Sub-scores were
achieved from PAID-5, PACIC-DSF, and WHO-5 (percentage of participants
suffering from depression, 8% vs. 13.8%) in Swiss DAWN2, compared to global average
scores. Weights are based on each country, as publicly
provided by each country's survey advisory group. Sample proportions are
depicted as points. The red point represents the mean value of the Swiss study
results, compared to the blue dot representing the entire sample of global
study data of people with diabetes.
PACIC-DSF Item 1: “I was asked how my
diabetes affects my life”, composite score
PACIC-DSF Item 12: “I was satisfied that
my care was well organised”, composite score.

Figure 3Scores achieved from SDSCA-6 in Swiss DAWN2, compared to global average scores. Weights
are based on each country, as publicly
provided by each country's survey advisory group. Sample means are depicted as
points, 95% confidence interval of the mean in brackets. The red point
represents the mean value of the Swiss study results, compared to the blue dot
representing the entire sample of global study data of people with diabetes.
Prescribed and recommended medication not included in Swiss sample.
Figure 4
illustrates the effects of HbA1c on the scores of completed questionnaires.

Figure 4Scores achieved from
EQ-5D-3L, PAID-5, SDSCA-6, PACIC-DSF, WHO-5 in Swiss DAWN2 depicting the
effect of “within-range” HbA1c (<7%) and “out-of-range” HbA1c (≥7%) on parameters
of the above mentioned questionnaires. A t-test has been used applying continuous
variables in
groups “within-range” and “out-of-range”.
Figure 5 illustrates the most commonly reported health concerns, figure 6
illustrates the distribution of the most commonly reported health complications,
and figure 7 illustrates the use of various types of diabetes self-management
education (DSME) sources in patients with type 1 diabetes or type 2 diabetes.

Figure 5Most common concerns
of patients with type 1 and type 2 diabetes .

Figure 6Most common health
complications of patients with type 1 and type 2 diabetes .
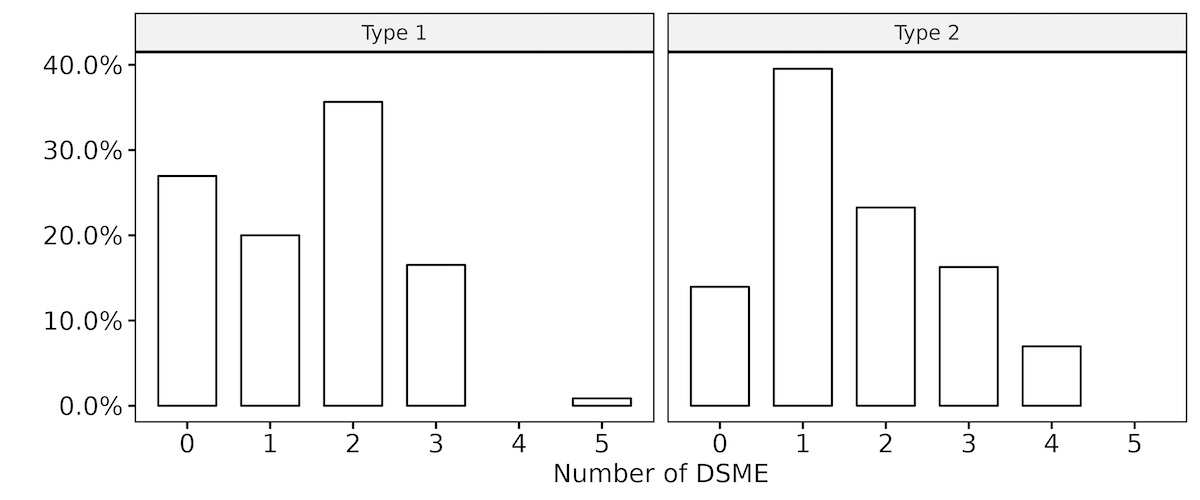
Figure 7Number of DSME sources used by patients with type 1 and type 2 diabetes at any time
prior to study
inclusion.
The EQ-5D-3L questionnaire
revealed a higher quality of life as compared to the global score (77.28 ±
16.73 vs. 69.3 ± 17.9, p <0.001, see figure 1). The PAID-5 questionnaire
indicated lower emotional distress in the Swiss study cohort as compared to the
global study cohort (22.28 ± 20.94 vs. 35.2
± 24.2, p <0.001, see figure 1). Hereby, patients with type 1 diabetes reported a
higher percentage of distress compared to those with type 2 diabetes (18.3 vs. 4.7,
p =
0.024). PACIC-DSF illustrated higher treatment-related and organisational
satisfaction in the Swiss study cohort as compared to the global study cohort
(60.3 ± 1.51 vs. 47.3 ± 24.3, p = 0.001, see figures 1 and 2). According to WHO-5,
well-being was significantly higher in the participants of the Swiss DAWN2 as
compared to the global score (71.38 ± 23.31 vs. 58 ± 13.8, p <0.001, see
figure 1), yet, depressive episodes were reported significantly less frequent
(8% vs. 13.8%, p = 0.043, see figure 2).
SDSCA-6 revealed higher frequencies
regarding the implementation of physical activity (4.40 ± 2.04 vs. 3.8 ± 2.7, p
<0.001), the self-measurement of blood glucose (6.43 ± 1.68 vs. 3.4 ± 2.8,
p <0.001), and higher compliance with recommended treatment strategies
(6.25 ± 1.92 vs. 3.2 ± 2.8, p < 0.001, see figure 3) in participants of the
Swiss DAWN2, compared to the global average. HbA1c
>7% correlated to higher emotional distress (PAID-5, 26.08 ± 23.37 vs.
18.80 ± 17.49, p = 0.024), less favourable diabetes-related eating habits (4.28
± 2.22 vs. 4.99 ± 2.15, p = 0.034) and lower frequencies of physical activity
(3.95 ± 2.16 vs. 4.72 ± 1.92, p = 0.014, see figure 4). A
total of 47.2% of the Swiss DAWN2 participants expressed at least one concern
related to their life with diabetes (see figure 5), including depressive mood
swings, concerns regarding future serious health complications, cognitive and
physical strength limitations, challenges in the management of health
complications, as well as to feel overwhelmed by diabetes and therapeutic
compliance, resulting in guilt. The most common concern among both diabetes
types involved the fear of future serious health complications. Patients with type
1 diabetes reported a higher level of concern regarding treatment-compliance and guilt
as
compared to patients with type 2 diabetes (2.53 ± 1.14 vs 1.82 ± 0.90, p = 0.001).
The most commonly
reported secondary complications related to diabetes were the following:
sleeping problems (35.6%), vision impairment (34.1%), depression (25.5%),
sexual impairment including erectile dysfunction (22.7%), and neurological
damages (17.4%). Patients with type 1 diabetes reported visual impairment (30.5%),
while patients with type 2 diabetes
reported sleeping problems/insomnia (46.3%) as the most common complication.
In total, 69.9% of respondents reported having at least one health-related
complication (see figure 6). A total of
28.8% of the Swiss DAWN2 participants had participated in DSME at any time
prior to study inclusion. Of those, 73% of patients with type 1 diabetes reported
using DSME sources
compared to 86% of patients with type 2 diabetes (see figure 7). A total of 161 participants
(67.4%)
reported using DSME tools for diabetes-specific education, data management or
physical training. A total of 85 participants (35.6%) had visited
diabetes-specific group training, 37 participants (15.5%) had attended online
diabetes programs and self-help support groups, 16 participants (6.7%) had
interacted in weight loss or sports courses, and 46 participants (19.2%) had
made use of regional offers provided by national diabetes associations.
Discussion
The Swiss DAWN2™ study illustrated higher scores for quality of life and treatment
satisfaction in individuals with diabetes compared to the global average. Diabetes-related
distress and the psychosocial burden of the disease were found to be lower in Switzerland
as compared to the global study cohort. Also, the prevalence of depression was lower
in the Swiss population.
As mentioned, a WHO-5 score ≤28 was associated with a higher probability of depression.
In total, 13.8% of the global cohort suffered from depression. In this regard, country-specific
comparisons, including the German and French study cohorts, reported values of 13.6%
and 14%, respectively [17, 18]. The Swiss cohort, on the other hand, shows a lower
prevalence of depression of only 8%. In addition, diabetes-related distress (PAID-5)
was also lower in the Swiss study cohort as compared to the global and French samples.
Approximately 30% of patients with diabetes suffer at least once in their lifetime
from clinically relevant depressive disorders, while 10% suffer from a major depressive
disorder at least once. Depression has a tremendous impact on glycaemic control and
significantly increases the risk for the acute and long-term complications of diabetes
[19, 20]. Concurrent depression has a negative impact on the quality of life in patients
with diabetes, reducing the implementation of physical activity and increasing the
demand for medical care and supervision [21, 22].
Swiss DAWN2™ indicated an overall positive relationship between patients and healthcare
experts in terms of support, organisational planning, and the acceptance of treatments
proposed by healthcare experts [23]. Insufficient time management and inadequate organisational
planning between health care professionals and patients may decrease glycaemic control
and diabetes management and also increase the time and financial expenses required
for successful treatment [24].
However, the present results also illustrate a potential to further support physical
activity and nutrition education in patients with diabetes. This is in line with global
observations indicating that the available single or group-based educational programs
may not be sufficiently advertised, leading to lower compliance and acceptance rates
for individuals with diabetes [25]. This situation may occur due to financial, time-wise,
and personnel constraints, even though the present study design does not allow for
the identification of factors leading to low perception rates.
Together, the original DAWN studies have shown that 48.8% of respondents participated
in DSME programs at least once. Therapeutic successes are largely dependent on patient’s
self-management behaviour (SMB), consisting of medication adherence, regular blood
glucose testing, behavioural adjustment, and treatment compliance [26]. Related research
also shows that diabetes-specific knowledge positively correlates to adequate SMB,
better glycaemic control, and improved HbA1c. In addition, structural DSME programs
and their availability are critical elements related to education and awareness-raising
in patients with diabetes [26].
As stated before, structured and unstructured DSME programs at the outpatient clinic
of the University Hospital of Bern were made available to all the study participants.
These programs intended to support patients with diabetes to adopt a healthy lifestyle,
thus preventing the occurrence and progression of diabetes-specific complications.
The long-term goals of diabetes rehabilitation programs are weight loss, healthier
lifestyle, motivational training, and accessibility to self-help groups in the diabetic
and cardiovascular field [27]. In this regard, 28.8% of the Swiss study cohort reported
using DSME, including diabetes training participation, while 27.1% of the cohort in
the neighbouring country of Germany reported using similar programs [17].
The present study is subject to certain limitations. First, the patient cohort was
recruited at a tertiary care center, which might limit the generalisability of the
present results. However, as the health care in Switzerland is coordinated nationwide,
differences from other Cantons might be present
but may not significantly inhibit access to diabetes care for patients and
caregivers. Importantly, it is necessary to extend the obtained knowledge to patients
treated outside of tertiary care centers. Second, eligibility was limited to patients
speaking one of the three main languages spoken nationwide (German, Italian, and French).
This inhibited the interpretation of the present results in patients with migration
backgrounds from regions where other languages are used; future studies must consider
this aspect. In summary, the present results cannot be generalised to the entire Swiss
population. Moreover, the internet methodology may not be fully representative due
to the possibility of systematic bias.
In summary, quality of life and treatment satisfaction were found to be favourable
in Switzerland as compared to global cohorts. Further, the coordination between patients
and health care providers seems sufficient, which strongly contributes to successful
diabetes management [28]. Access to educational programs such as individual, group-based,
or paper-based initiatives should be foregrounded to strengthen a diabetic patient’s
educational level and promote a stable and healthy lifestyle [29, 30].
The findings of this Swiss DAWN2™ study may help identify the unmet needs in the treatment
of diabetes and may also facilitate better self-management and psychological support,
ultimately reducing the disease burden on people with diabetes. Further studies in
this field are essential, including but not limited to family members, general practitioners,
and caregivers, in order to extend the present results’ generalisability and comparability.
Prof. Dr.
med. Markus Laimer
Department
of Diabetes, Endocrinology, Nutritional Medicine and Metabolism
Inselspital
Bern, University Hospital and University of Bern
Freiburgstrasse
15
CH-3010 Bern
markus.laimer[at]insel.ch
References
1. Barnard KD, Peyrot M, Holt RI. Psychosocial support for people with diabetes: past,
present and future. Diabet Med. 2012 Nov;():1358–60. https://doi.org/10.1111/j.1464-5491.2012.03727.x
2. Skovlund SE, Peyrot M; DAWN International Advisory Panel. The Diabetes Attitudes,
Wishes, and Needs (DAWN) program: a new approach to improving outcomes of diabetes
care. Diabetes Spectr. 2005 Jul;():136–42. https://doi.org/10.2337/diaspect.18.3.136
3. World Health Organization. Diabetes [Internet]. World Health Organization; 2022. https://www.who.int/health-topics/diabetes#tab=tab_1
4. Bădescu SV, Tătaru C, Kobylinska L, Georgescu EL, Zahiu DM, Zăgrean AM, et al. The
association between Diabetes mellitus and Depression. J Med Life. 2016;():120–5.
5. Bajor LA, Gunzler D, Einstadter D, Thomas C, McCormick R, Perzynski AT, et al. Associations
between comorbid anxiety, diabetes control, and overall medical burden in patients
with serious mental illness and diabetes. Int J Psychiatry Med. 2015;():309–20. https://doi.org/10.1177/0091217415589307
6. Dybdal D, Tolstrup JS, Sildorf SM, Boisen KA, Svensson J, Skovgaard AM, et al. Increasing
risk of psychiatric morbidity after childhood onset type 1 diabetes: a population-based
cohort study. Diabetologia. 2018 Apr;():831–8. https://doi.org/10.1007/s00125-017-4517-7
7. Gandhi KK, Baranowski T, Anderson BJ, Bansal N, Redondo MJ. Psychosocial aspects of
type 1 diabetes in Latino- and Asian-American youth. Pediatr Res. 2016 Sep;():347–55.
https://doi.org/10.1038/pr.2016.87
8. Delamater AM, Jacobson AM, Anderson B, Cox D, Fisher L, Lustman P, et al.; Psychosocial
Therapies Working Group. Psychosocial therapies in diabetes: report of the Psychosocial
Therapies Working Group. Diabetes Care. 2001 Jul;():1286–92. https://doi.org/10.2337/diacare.24.7.1286
9. Rechenberg K, Whittemore R, Grey M. Anxiety in youth with type 1 diabetes. J Pediatr
Nurs. 2017;:64–71. https://doi.org/10.1016/j.pedn.2016.08.007
10. Rubin RR, Peyrot M, Siminerio LM; International DAWN Advisory Panel. Health care and
patient-reported outcomes: results of the cross-national Diabetes Attitudes, Wishes
and Needs (DAWN) study. Diabetes Care. 2006 Jun;():1249–55. https://doi.org/10.2337/dc05-2494
11. Peyrot M, Burns KK, Davies M, Forbes A, Hermanns N, Holt R, et al. Diabetes Attitudes
Wishes and Needs 2 (DAWN2): a multinational, multi-stakeholder study of psychosocial
issues in diabetes and person-centred diabetes care. Diabetes Res Clin Pract. 2013 Feb;():174–84.
https://doi.org/10.1016/j.diabres.2012.11.016
12. Nicolucci A, Kovacs Burns K, Holt RI, Comaschi M, Hermanns N, Ishii H, et al.; DAWN2
Study Group. Diabetes Attitudes, Wishes and Needs second study (DAWN2™): cross-national
benchmarking of diabetes-related psychosocial outcomes for people with diabetes. Diabet
Med. 2013 Jul;30(7):767–77. https://doi.org/10.1111/dme.12245
13. Iturralde E, Rausch JR, Weissberg-Benchell J, Hood KK. Diabetes-related emotional
distress over time. Pediatrics. 2019 Jun;():e20183011. https://doi.org/10.1542/peds.2018-3011
14. Peter P, Lipska K. The rising cost of diabetes care in the USA. Lancet Diabetes Endocrinol.
2016 Jun;():479–80. https://doi.org/10.1016/S2213-8587(15)00519-7
15. Mansell K, Perepelkin J. Patient awareness of specialized diabetes services provided
in community pharmacies. Res Social Adm Pharm. 2011 ;():396–405. https://doi.org/10.1016/j.sapharm.2010.10.004
16. CoreTeam. R: A language and environment for statistical computing. R Foundation for
Statistical Computing, Vienna, Austria. Available from: http://www.R-project.org/. 2013
17. Kulzer B, Lüthgens B, Landgraf R, Hermanns N. Diabetesbezogene Belastungen, Wohlbefinden
und Einstellung von Menschen mit Diabetes. Diabetologe (Heidelb). 2015 May;():211–8.
https://doi.org/10.1007/s11428-015-1335-8
18. Reach G, Consoli SM, Halimi S, Colas C, Duclos M, Fontaine P, et al. The multinational
second Diabetes, Attitudes, Wishes and Needs study: results of the French survey.
Patient Prefer Adherence. 2015 Feb;:289–97. https://doi.org/10.2147/PPA.S68941
19. Nouwen A, Adriaanse MC, van Dam K, Iversen MM, Viechtbauer W, Peyrot M, et al.; European
Depression in Diabetes (EDID) Research Consortium. Longitudinal associations between
depression and diabetes complications: a systematic review and meta-analysis. Diabet
Med. 2019 Dec;():1562–72. https://doi.org/10.1111/dme.14054
20. Group TE; EuroQol Group. EuroQol—a new facility for the measurement of health-related
quality of life. Health Policy. 1990 Dec;():199–208. https://doi.org/10.1016/0168-8510(90)90421-9
21. Kalra S, Jena BN, Yeravdekar R. Emotional and psychological needs of people with diabetes.
Indian J Endocrinol Metab. 2018;():696–704. https://doi.org/10.4103/ijem.IJEM_579_17
22. Lustman PJ, Anderson RJ, Freedland KE, de Groot M, Carney RM, Clouse RE. Depression
and poor glycemic control: a meta-analytic review of the literature. Diabetes Care.
2000 Jul;():934–42. https://doi.org/10.2337/diacare.23.7.934
23. Peyrot M, Rubin RR, Lauritzen T, Snoek FJ, Matthews DR, Skovlund SE. Psychosocial
problems and barriers to improved diabetes management: results of the Cross-National
Diabetes Attitudes, Wishes and Needs (DAWN) Study. Diabet Med. 2005 Oct;():1379–85.
https://doi.org/10.1111/j.1464-5491.2005.01644.x
24. Fiore, V., Marci, M., Poggi, A., Giagulli, V., Licchelli, B., Iacoviello, M., Guastamacchia,
E., De Pergola, G. and Triggiani, V., 2021. The association between diabetes and depression:
a very disabling condition.
25. Moulton CD, Pickup JC, Ismail K. The link between depression and diabetes: the search
for shared mechanisms. Lancet Diabetes Endocrinol. 2015 Jun;():461–71. https://doi.org/10.1016/S2213-8587(15)00134-5
26. Beck J, Greenwood DA, Blanton L, Bollinger ST, Butcher MK, Condon JE, et al.; 2017
Standards Revision Task Force. 2017 National standards for diabetes self-management
education and support. Diabetes Care. 2017 Oct;():1409–19. https://doi.org/10.2337/dci17-0025
27. Diafit i. DIAfit, Diabetes mellitus, Rehabilitation, Selbsthilfegruppe, Therapie,
Ausbildung, Diabetes-Gesellschaft, SDG, SGED [Internet]. Diafit.ch. 2022. Available
from: http://www.diafit.ch/
28. Beaser RS, Okeke E, Neighbours J, Brown J, Ronk K, Wolyniec WW. Coordinated primary
and specialty care for type 2 diabetes mellitus, guidelines, and systems: an educational
needs assessment. Endocr Pract. 2011;():880–90. https://doi.org/10.4158/EP10398.OR
29. Mosnier-Pudar H, Hochberg G, Eschwege E, Virally ML, Halimi S, Guillausseau PJ, et
al. How do patients with type 2 diabetes perceive their disease? Insights from the
French DIABASIS survey. Diabetes Metab. 2009 Jun;():220–7. https://doi.org/10.1016/j.diabet.2009.02.001
30. Nassar CM, Montero A, Magee MF. Inpatient diabetes education in the real world: an
overview of guidelines and delivery models. Curr Diab Rep. 2019 Sep;():103. https://doi.org/10.1007/s11892-019-1222-6






